Department of Anatomy (School
of Medicine) &
Department of Zoology
(College of Science)
B.S., Biological Science, Purdue, 1970
Ph.D., Neuroscience, UCSD, 1975
NIH Postdoctoral Fellow , Yale, 1976-77
Neurobiology
Investigation of Axonal Diameter (1986)
Flies are hard to swat. The sudden stimulus of an approaching fly-swatter often startles the fly, leading to a quick escape. In many small creatures, including animals as diverse as flies, earthworms, crayfish, and squid, reflexive startle responses are triggered by signals sent along exceptionally large nerve fibers [1,3,4,5,6]. These large fibers, called "giant axons", seem to be an evolutionary adaptation for survival. Their extreme size lowers electrical resistance while increasing power, permitting especially fast and reliable signalling. Fast and reliable signals, in turn, promote quick responses so the startled animal can escape from danger [2]. The giant axons which enable many fly species to dodge the fly swatter carry signals to the muscles which are used to begin flight.
People and other mammals do not have giant axons, even though nerve impulses from brain to muscle must travel a much greater distance than in flies. In fact, the largest axons in humans are much thinner than those in flies or earthworms. Of course, speed is still a virtue even for large animals like ourselves. But instead of increasing conduction velocity by increasing axonal diameter (which decreases internal axonal resistance), our nerve fibers depend on myelin, a fatty covering which lowers membrane capacitance. This increases conduction velocity by allowing minute electrical currents to travel much more efficiently, essentially by "skipping" along the length of a nerve fiber.
But even though giant axons are not found in human bodies, giant axons have proven quite useful for neurobiologists. Decades ago, giant axons from squids were used to elucidate the basic physics of nerve impulse conduction. A convenient laboratory demonstration involves giant axons in earthworms, which produce electrical signals powerful enough to be recorded from outside the intact animal [3]. Giant axons in crayfish, cockroaches and locusts have been used to study neural circuitry and synaptic activity [4,5,6]. More recently, mutations affecting the giant axons in laboratory fruit flies have been used to investigate how genes influence the form and function of nerve cells.
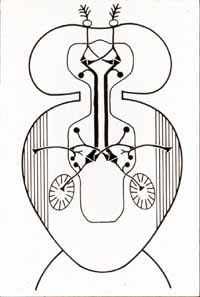 Instinctive
behaviors like the fly's startle response depend on the functional properties
of individual nerve cells. These properties, in turn, are shaped by
the DNA molecules contained within each living cell. Biologists can
explain how DNA is translated into proteins but have yet to learn how the
genetic material informs more complex patterns of biological organization,
including such intricate details as the specific size and shape of individual
nerve cells. Flies provide convenient subject for research into such
problems, because they are freely available and inexpensive to maintain in
the laboratory. The large size of their giant axons makes these highly
specialized nerve fibers comparatively easy to study. Dr. King, together
with colleagues at Yale University and elsewhere, has described the morphology
of giant axons and related nerve cells within the nervous system of Drosophila
melanogaster, the laboratory fruit fly [7]. Behavior
mutants, selected for defects in the startle response which is controlled
by these cells, have proven useful for neurogenetic study [8].
Instinctive
behaviors like the fly's startle response depend on the functional properties
of individual nerve cells. These properties, in turn, are shaped by
the DNA molecules contained within each living cell. Biologists can
explain how DNA is translated into proteins but have yet to learn how the
genetic material informs more complex patterns of biological organization,
including such intricate details as the specific size and shape of individual
nerve cells. Flies provide convenient subject for research into such
problems, because they are freely available and inexpensive to maintain in
the laboratory. The large size of their giant axons makes these highly
specialized nerve fibers comparatively easy to study. Dr. King, together
with colleagues at Yale University and elsewhere, has described the morphology
of giant axons and related nerve cells within the nervous system of Drosophila
melanogaster, the laboratory fruit fly [7]. Behavior
mutants, selected for defects in the startle response which is controlled
by these cells, have proven useful for neurogenetic study [8].
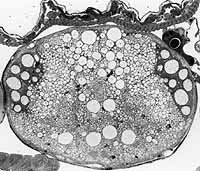 Dr.
King is also comparing the size distribution of axons from various other species
of flies (insect order Diptera), taking advantage of "natural experiments"
in genetic change that have been provided by evolution. The presence
or absence of particular giant axons reflects the influence of mutation and
natural selection over the history of each species [Figure
and Abstract]. By comparing how specific characteristics of individual
nerve cells have adapted to varied circumstances, Dr. King seeks to understand
genetic flexibility and evolutionary versatility.
Since there must be some limit to the adaptive detail that can be encoded
in animal's genes, there must also be some limit to the freedom with which
adaptations can evolve in any species. Genes cannot possibly contain
enough information to specifiy every property of every nerve cell individually.
A fruit fly has only a few thousand genes, but its nervous system contains
tens of thousands of nerve cells. Yet some individual nerve cells nevertheless
do display unique adaptive traits, such as giant axons.
Dr.
King is also comparing the size distribution of axons from various other species
of flies (insect order Diptera), taking advantage of "natural experiments"
in genetic change that have been provided by evolution. The presence
or absence of particular giant axons reflects the influence of mutation and
natural selection over the history of each species [Figure
and Abstract]. By comparing how specific characteristics of individual
nerve cells have adapted to varied circumstances, Dr. King seeks to understand
genetic flexibility and evolutionary versatility.
Since there must be some limit to the adaptive detail that can be encoded
in animal's genes, there must also be some limit to the freedom with which
adaptations can evolve in any species. Genes cannot possibly contain
enough information to specifiy every property of every nerve cell individually.
A fruit fly has only a few thousand genes, but its nervous system contains
tens of thousands of nerve cells. Yet some individual nerve cells nevertheless
do display unique adaptive traits, such as giant axons.
By learning how the formation of specialized individual cells can be guided by information in genes and chromosomes, and by analyzing the constraints upon the evolution of such detailed cellular traits, Dr King hopes for clues for underlying principles of neural and genetic organization.
Also see: Comparative neurobiology 2013 essay / 2006 essay / 1997 essay
1. T.H. Bullock (1984) Comparative neuroethology of startle, rapid escape, and giant fiber-mediated responses. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
2. M.V.L. Bennet (1984) Escapism: Some startling revelations. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
3. C.D. Drewes (1984) Escape reflexes in earthworms and other annelids. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
4. R.E. Ritzmann (1984) The cockroach escape response. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
5. R.G. Pierson and M. O'Shea (1984) Escape behavior of the locust: The jump and its initiation by visual stimuli. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
6. F.B. Krasne and J.J. Wine (1984) The production of crayfish tailflip escape responses. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
7. R.J. Wyman, J.B. Thomas, L. Salkoff and D. King (1984) The Drosophila giant fiber system. In: Neural Mechanisms of Startle Behavior, R. Eaton, ed. Plenum Press, New York.
8. R.J. Wyman, J.B. Thomas, L. Salkoff and W. Costello (1985) The Drosophila thorax as a model system for neurogenetics. In: Model Neural Networks and Behavior, A. Selverston, ed. Plenum Press, New York.
